Video via TK News
Transcript: Tim Robbins and the Lost Art of Finding Common Ground
Just saw The Shawshank Redemption, for the billionth time, yesterday.
Video via TK News
Transcript: Tim Robbins and the Lost Art of Finding Common Ground
Just saw The Shawshank Redemption, for the billionth time, yesterday.
Over the weekend, a local news outlet in Tucson, Arizona, reported that a 14-year-old girl had been denied her medication, methotrexate, for her debilitating arthritis and osteoporosis, because methotrexate can possibly induce a miscarriage and the girl is of childbearing age. This comes just after Arizona’s pre-Civil War, total abortion ban took effect at the end of last month.
14-Year-Old Speaks Out After Being Denied Medication Because She’s Childbearing Age

In 1986 Congress passed the National Childhood Vaccine Injury Act (NCVIA), creating a no-fault compensation program to stabilize a vaccine market adversely affected by an increase in vaccine-related lawsuits and to facilitate compensation to claimants who found pursuing legitimate vaccine-inflicted injuries too difficult and cost prohibitive.
Why vaccine manufacturers are exempt from liability
The move came after a report on fake US military-linked accounts that were promoting pro-Western narratives on Facebook and Twitter
Pentagon Opens Review Of Its Clandestine Psychological Operations
Related:
Pentagon Opens Sweeping Review of Clandestine Psychological Operations
With the rise of Russia and China as strategic competitors, military commanders have wanted to fight back, including online. And Congress supported that. Frustrated with perceived legal obstacles to the Defense Department’s ability to conduct clandestine activities in cyberspace, Congress in late 2019 passed a law affirming that the military could conduct operations in the “information environment” to defend the United States and to push back against foreign disinformation aimed at undermining its interests. The measure, known as Section 1631*, allows the military to carry out clandestine psychologic operations without crossing what the CIA has claimed as its covert authority, alleviating some of the friction that had hindered such operations previously.
VERIFY: New COVID booster approved from final study of mice, not humans
According to documents on the CDC website, human tests of Moderna’s version of the new booster are still “ongoing.” Right now, there is only final evidence from “8-10 mice” per group.”
The FDA’s website shows for Pfizer the agency also relayed on the final study evidence from “8 mice.” But human trials are also ongoing. So far, the new boosters “showed a similar local reaction and systemic event profile.” That means side effects appear to be about the same including “mild to moderate injection site pain, fatigue, and muscle pain.”
…
“To advance the Omicron BA.4/BA.5 bivalent vaccine as rapidly as possible, regulators, including the FDA, WHO and EMA, have advised that our submissions be based on safety and immunogenicity data generated in adults with an Omicron BA.1 bivalent vaccine and supported by BA.4/BA.5 bivalent pre-clinical data and BA.4/BA.5 bivalent quality and manufacturing data. These packages follow a similar model to that provided to public health officials who evaluate seasonal flu vaccines each year. Moving forward, this approach may help address the pressing need for vaccines that provide a high level of protection against current and emerging variants of concern so that we can remain vigilant against this evolving virus. The FDA has authorized our Omicron BA.4/BA.5 bivalent vaccine based on clinical data generated in adults with an Omicron BA.1 bivalent vaccine, pre-clinical data with a BA.4/BA.5 bivalent vaccine and BA.4/BA.5 bivalent quality and manufacturing data. Pre-clinical data showed a booster dose of Pfizer and BioNTech’s Omicron BA.4/BA.5- bivalent vaccine generated a strong neutralizing antibody response against Omicron BA.1, BA.2 and BA.4/BA.5 variants, as well as the original wild-type strain. A clinical study investigating the safety, tolerability and immunogenicity of the Omicron BA.4/BA.5 bivalent vaccine in individuals 12 years of age and older is ongoing. Data will be shared with regulators when available. We are facing a virus with an exceptionally high mutation rate, which the nimble mRNA platform is well situated to address. Only the mRNA sequence requires updating to match emerging strains, and utilizing a bivalent vaccine aims to provide broader coverage against known and future COVID-19 variants of concern.”
Related:
Read More »Americans Can Now Expect to Live Three Years Less than Cubans
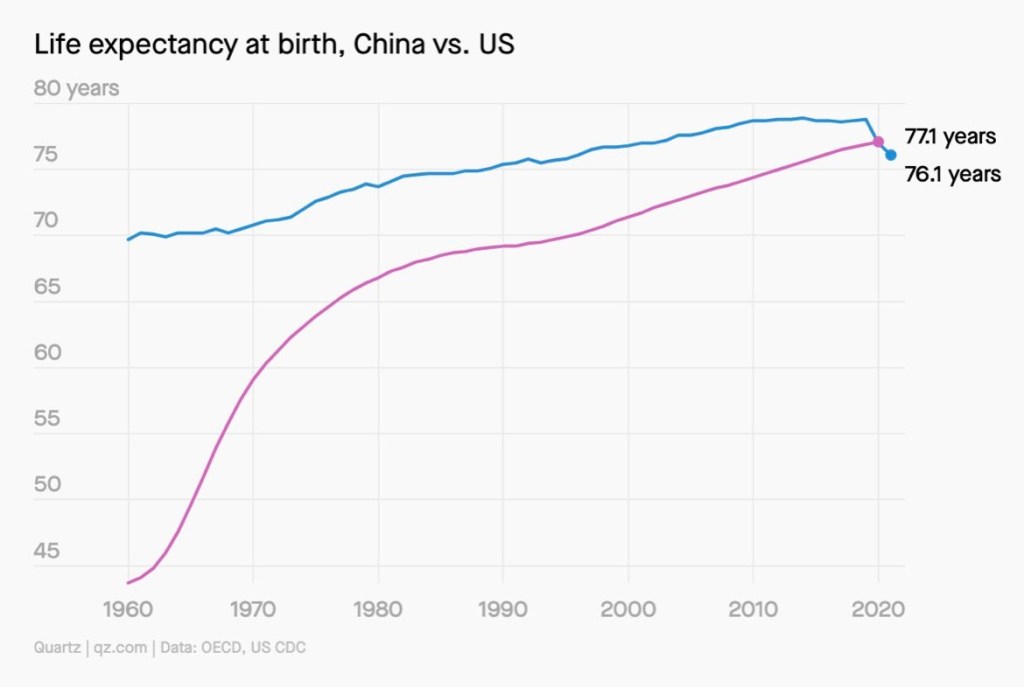
The decline means that several countries have caught up with or surpassed the U.S. According to World Bank data, Cuba’s life expectancy is 78.9, and China’s is 77.1, although both figures are from 2020 rather than 2021.
Other countries that are ahead of the U.S. in terms of life expectancy include: Colombia, Uruguay and Chile; Costa Rica, Panama and Puerto Rico; and Turkey, Greece and Albania.
…
Cubans have access to free healthcare, which is one of the key socialist principles. The country has a high ratio of medical professionals and focuses on prevention and primary care.
Related:
By Chris Hedges
The U.S. is not officially at war with Russia — but our war economy demands conformity and suppresses dissent
Ukraine, media censorship and the ruthless politics of permanent war
Researchers find that the nation had become an outlier among other rich countries in mortality rates long before the pandemic—and that Americans are dying younger than their peers abroad.
United States of Death? Study Shows Worrying Mortality Rates of Broken Health System
Pfizer’s anti-COVID pill Paxlovid shows no benefit for younger adults
The report’s authors found that Pfizer’s antiviral medication Paxlovid offered little to no benefit for younger adults. However, it did reduce the risk of hospitalization for high-risk seniors. Notably, supplementary material from the original study of Paxlovid in high-risk non-hospitalized adults with COVID-19 during the Delta wave had demonstrated benefits in those younger than 65, albeit the difference compared to the placebo was much less than in those 65 and older.
…
Among those over 65, there was a 73 percent decrease in the hospitalization rate and a 79 percent reduction in the risk of death. However, patients between the ages of 40 and 65 saw no benefit in taking the antiviral medication in either category, regardless of previous immunity status.
…
Another critical study from Hong Kong published in Lancet Infectious Diseases on the same day as the Israeli study but which went unmentioned in the press offered further evidence of Paxlovid’s limited therapeutic role. The authors reviewed their clinical experience with Paxlovid and Lagevrio, Merck’s antiviral pill, Molnupiravir, in hospitalized patients. They compared them to hospitalized patients who did not receive those medications during the horrific wave of infections that slammed into the semi-autonomous region in February and March.
…
The mortality risk reduction for Lagevrio was 52 percent, and for Paxlovid it was 66 percent. Those receiving antivirals had a lower risk of their disease progressing, but the drugs did not significantly impact their need for mechanical ventilation or ICU admission. The patients in the study averaged in age from mid-70s to early 80s.
…
Given the results of these studies, it bears mentioning that the Centers for Disease Control and Prevention (CDC) has recently estimated that approximately 95 percent of Americans aged 16 and older have some level of immunity against COVID-19.
4yo Boy Kicked Out of School, Police Called — Because He Wasn’t Wearing a Mask
Highlighting the utter insanity of these policies is the fact that the very next day, the district switched their stance. The day before, it was illegal to enter the school without a mask but now, 24 hours later, it was no longer illegal.
Even the WHO says children under 5 don’t need to wear masks!
You must be logged in to post a comment.